 
It also reacts violently with water, spewing corrosive liquid. Potassium metal is very dangerous and can form an explosive coating if it is kept in air. A few good sources of potassium are bananas, apricots and raisins. This is needed for muscles to contract (get shorter and move things.) It is needed for the heart to beat (push blood through blood vessels.) If the potassium level in the blood is too high or too low it can cause death because the heart stops. This means go from a negative to a positive electrical charge. It helps biological membranes depolarize. The ions send messages from cells to other cells. That is why fertilizers have potassium compounds in them. 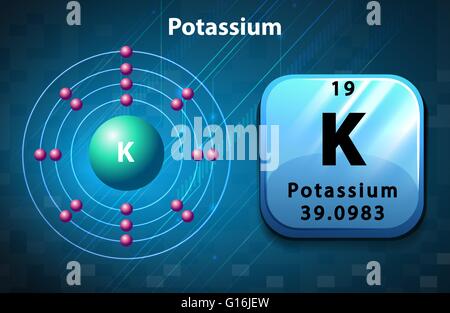
Potassium ions are very important to organisms. Potassium compounds are used in soap, fertilizer, explosives, and matches. It is also used in some scientific instruments. Potassium metal is used to absorb water from solvents. The potassium hydroxide or potassium chloride has to be melted at a very high temperature. It is extracted from them by electrolysis of potassium hydroxide or potassium chloride. Potassium does not occur in nature because it is too reactive. It is used to make fertilizer, soap, and glass. In past centuries potash was made from ashes in pots. Potash is a mixture of potassium carbonate and potassium hydroxide that has been used for a very long time. The word potassium comes from the word "potash". Potassium sulfate, colorless, used in fertilizers.Potassium sulfite, used in food preservation.Potassium permanganate, purple, disinfectant, oxidizing agent.Potassium perchlorate, used in some rockets.Potassium manganate, used to make potassium permanganate.Potassium iodide, used to supply iodine.Potassium iodate, used to supply iodine.Potassium hypomanganate, bright blue hypomanganate, rare.Potassium hydroxide, also known as potash, caustic, strong base, white solid.Potassium fluoride, used to make fluorine, corrosive.Potassium dichromate, red solid, oxidizing agent, toxic.Potassium chromate, yellow solid, oxidizing agent, toxic.Potassium chlorate, used in matches and explosives.Potassium carbonate, colorless, reacts with acids.Potassium bromide, colorless, used as sedative.Potassium bromate, colorless, oxidizing agent, used in flour.Potassium arsenite, colorless solid, toxic.Potassium arsenate, oxidizing agent, toxic.Potassium chromate is toxic because of the chromate, not the potassium. Potassium chromate is colored because of the chromate, not the potassium. If they are toxic, it is because of the anion. Potassium hydroxide is used in the electrolyte of alkaline cells. Potassium chloride can be used as a substitute for table salt. Potassium ions are colorless and similar to sodium ions. Potassium compounds are only in one oxidation state: +1. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
